
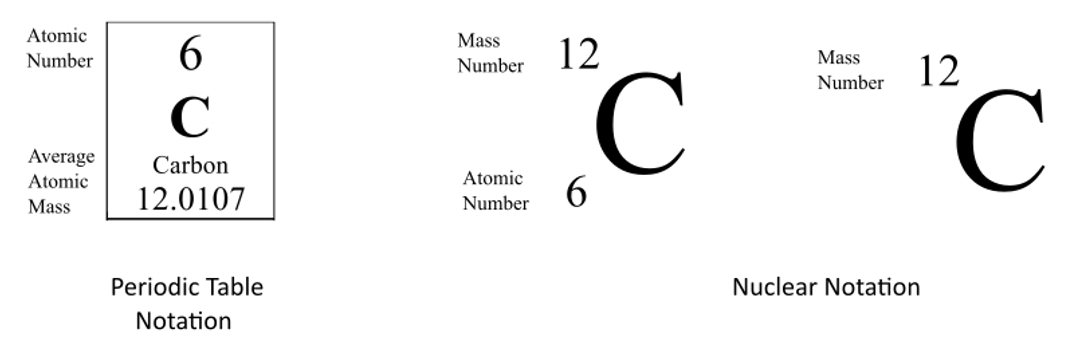
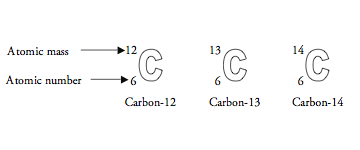
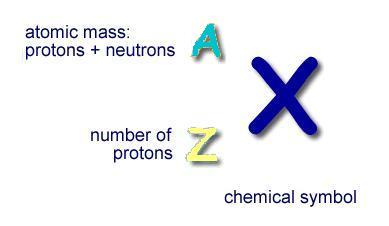
Atomic notation X A Z X = Symbol (C, Au) A = Atomic Mass Number = #nucleons (Protons + Neutrons) Z = Atomic Number = #protons C 12 6 Carbon A = 12, Z = - ppt download

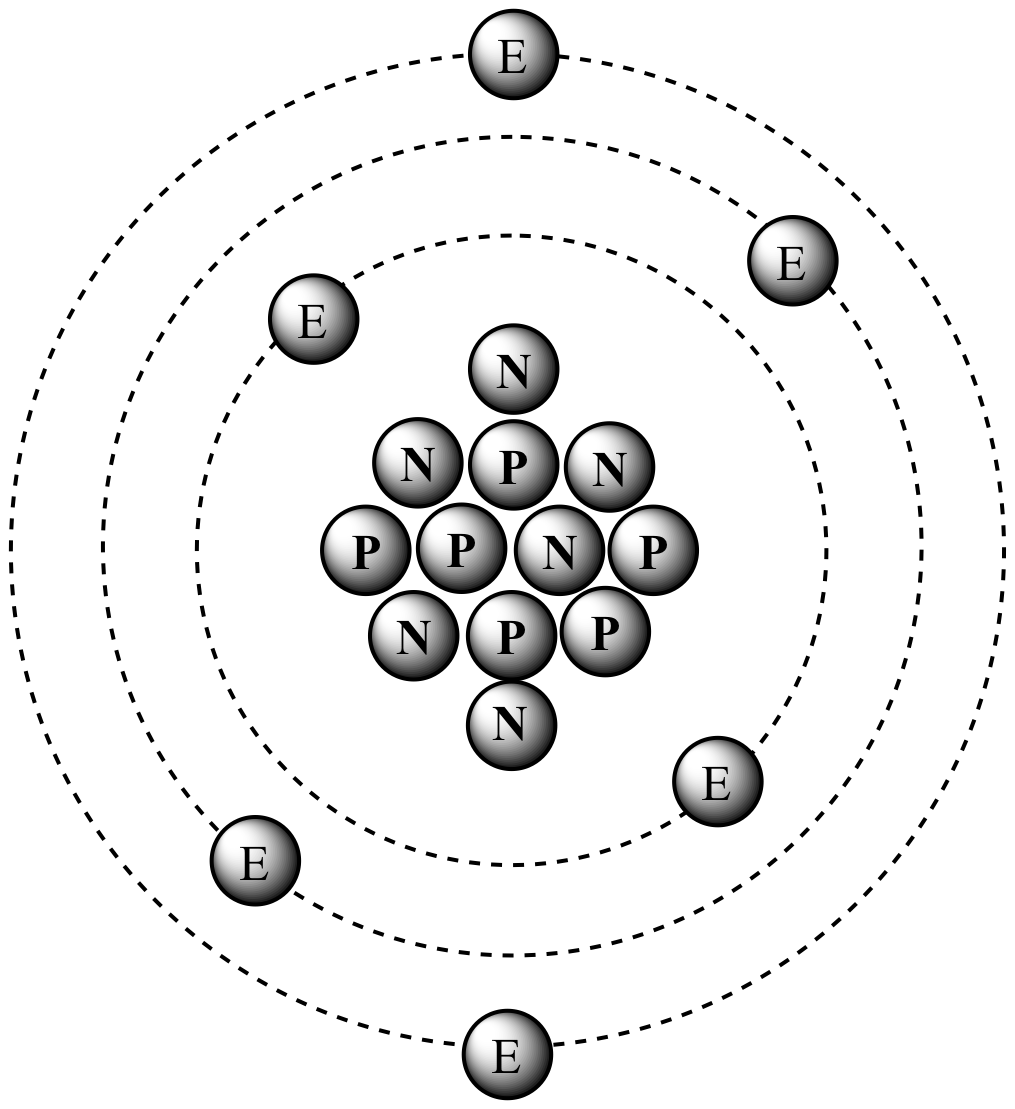
Refer to the diagram below to answer the following questions. What is the Atomic Number and Atomic - Brainly.in
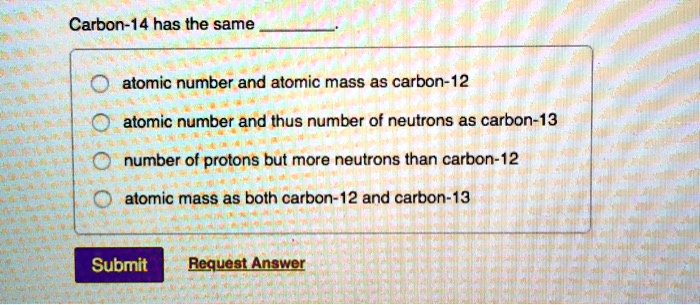
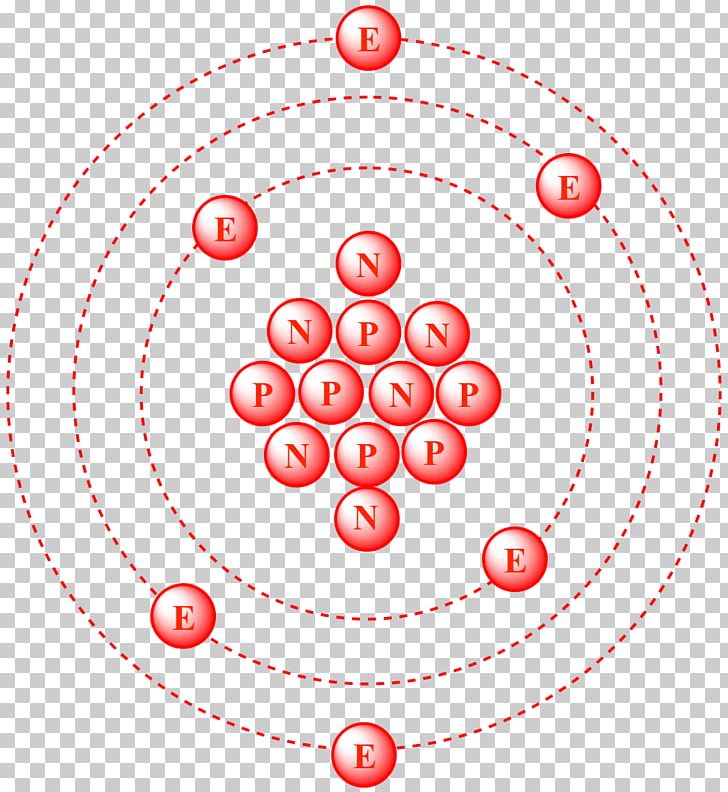
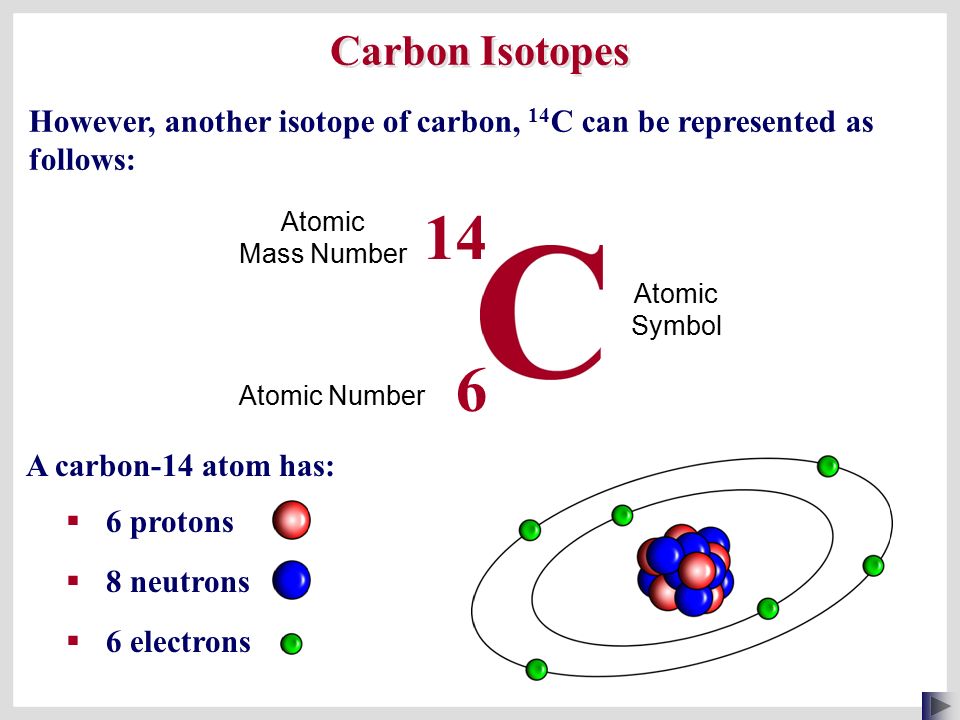
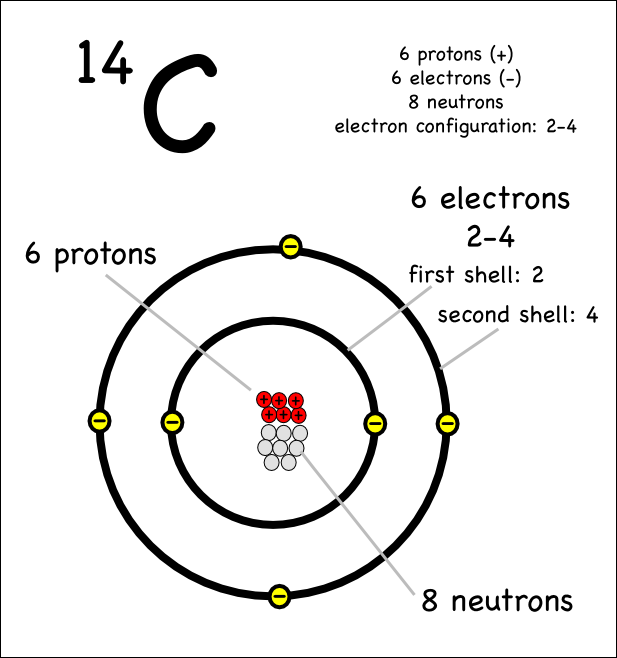
SOLVED: Carbon-14 has the same atomic number and atomic mass as carbon-12 atomic number and thus number of neutrons as carbon-13 number of protons but more neutrons than carbon-12 atomic mass as

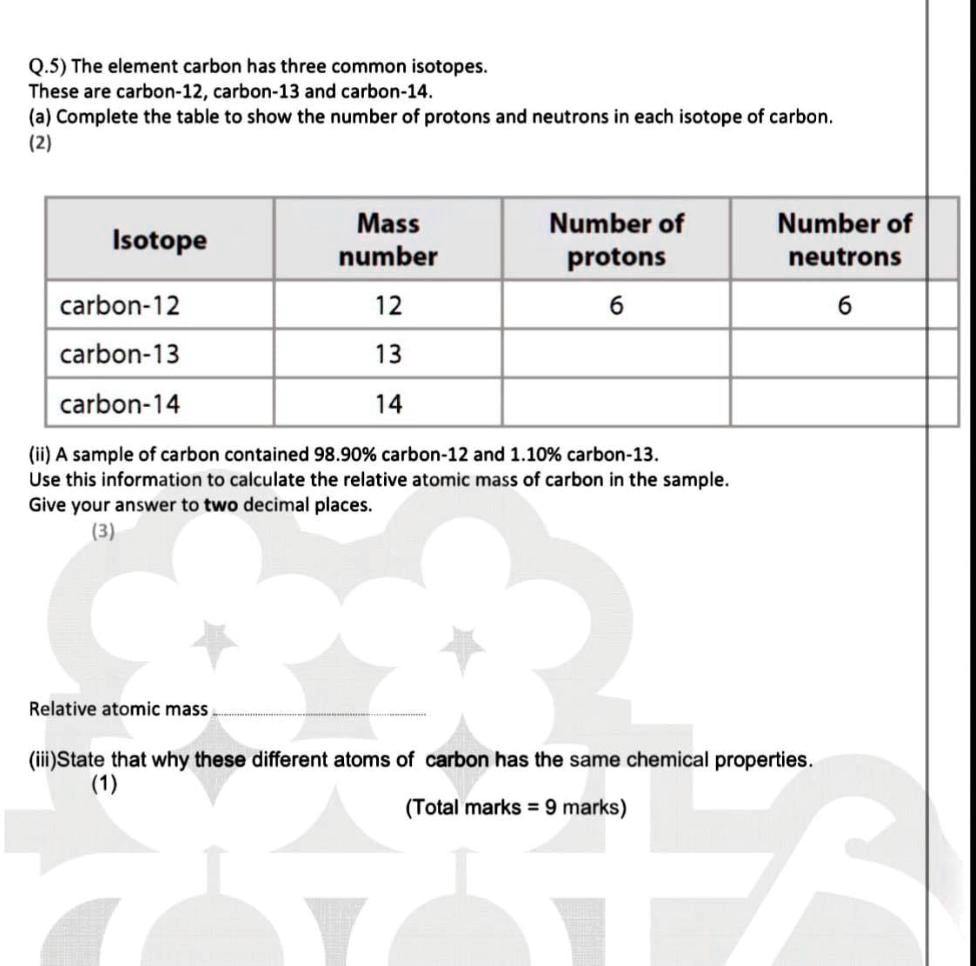
Atomic Number, Isotopes and Isobars - Definition, Examples And FAQS of Atomic Number, Isotopes and Isobars.






:max_bytes(150000):strip_icc()/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)





