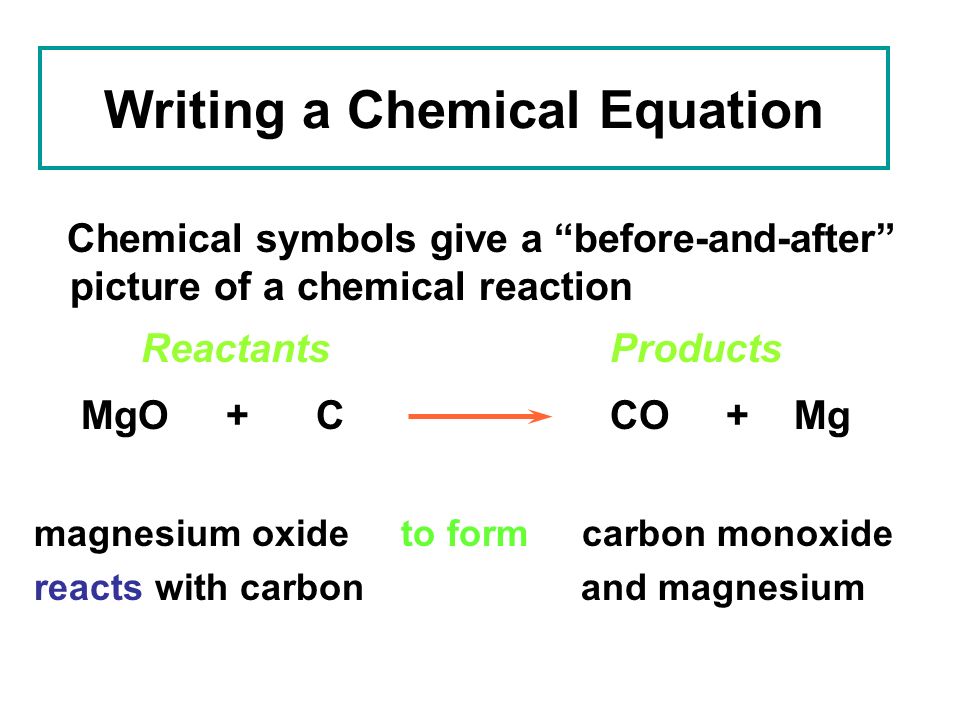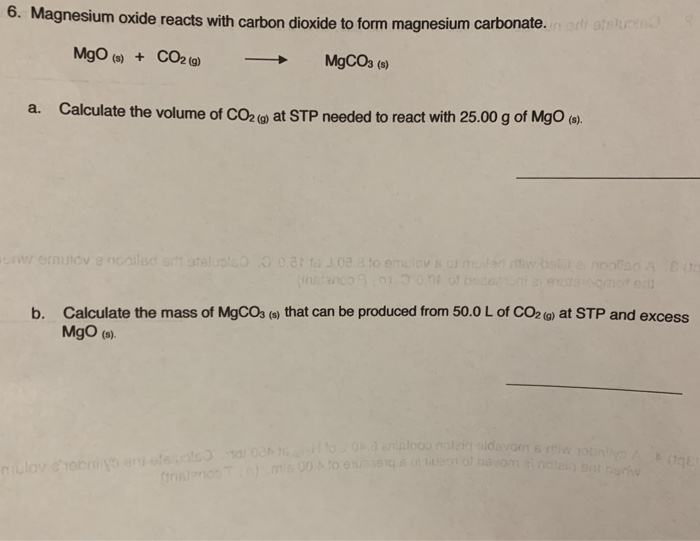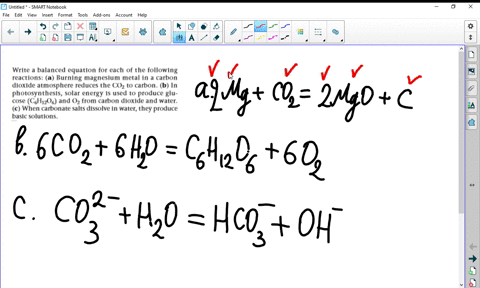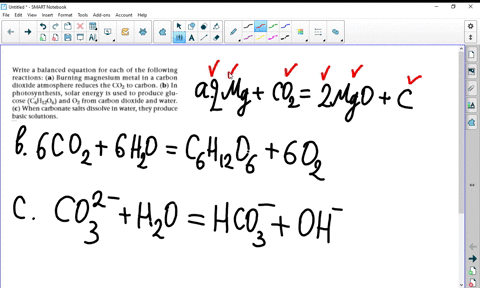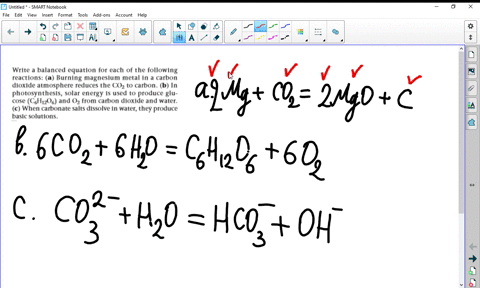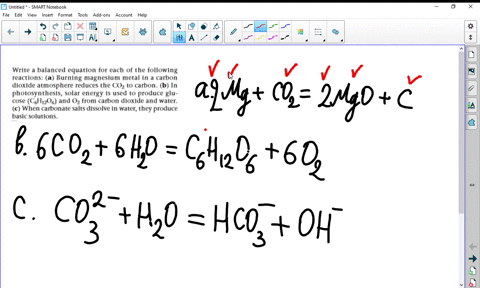
SOLVED:Write a balanced equation for each of the following reactions: (a) Burning magnesium metal in a carbon dioxide atmosphere reduces the CO2 to carbon. (b) In photosynthesis, solar energy is used to
Magnesium and carbon dioxide – Student sheet Burning magnesium in carbon dioxide – what will happen? To do
Magnesium and carbon dioxide – Student sheet Burning magnesium in carbon dioxide – what will happen? To do

Write the balanced chemical equation for the following reaction Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in an acidic medium

20.0 mg of a magnesium carbonate sample decomposes on heating to give carbon dioxide and 8.0 mg magnesium oxide. What will be the percentage purity of magnesium carbonate in the sample? [Atomic

Magnesium-Promoted Rapid Nucleation of Carbon Dioxide Hydrates | ACS Sustainable Chemistry & Engineering
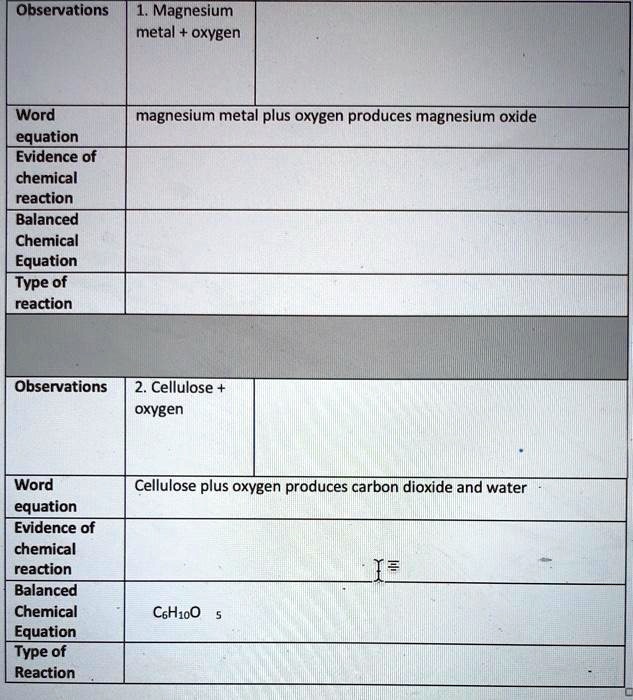
SOLVED: Observations Magnesium metal oxygen Word equation Evidence of chemical reaction Balanced Chemical Equation Type of reaction magnesium metal plus oxygen produces magnesium oxide Observations 2. Cellulose oxygen Word equation Evidence of

Metal carbonates are known to undergo thermal decomposition, producing the metal oxide and releasing carbon dioxide. The process is described by the following generic equation, in which M represents an unknown divalent

20.0 mg of a magnesium carbonate sample decomposes on heating to give carbon dioxide and 8.0 mg magnesium oxide. What will be the percentage purity of magnesium carbonate in the sample? [Atomic