5 72 grams of hydrated sodium carbonate is heated strongly The white residue left over is 2 12 grams Calculate - Chemistry - - 12355542 | Meritnation.com
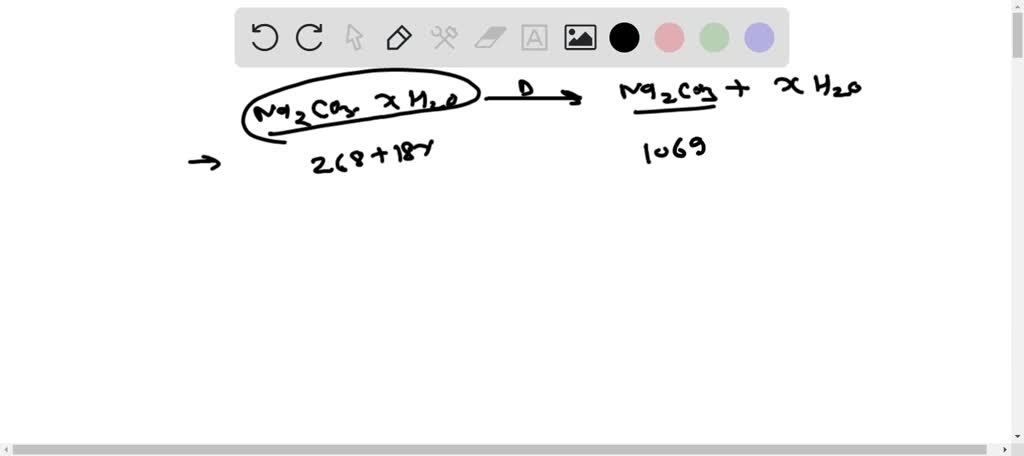
SOLVED: The molar mass of hydrated sodium carbonate is 268g.mol Calculate the number of moles of water of crystalisation (x)If the formula hydrated sodium carbonate is Na2Co3 .xH2O
1 The energy level diagram for the reaction between sodium hydrogen carbonate and dilute hydrochloric acid is shown. sodium chlo

SOLVED: Calculate the formula of hydrated sodium carbonate given the following data. Mass of crucible Mass of crucible and hydrate Mass Of crucible and anhydride (NaCO ) 17.76 5162 4 30.30 g (
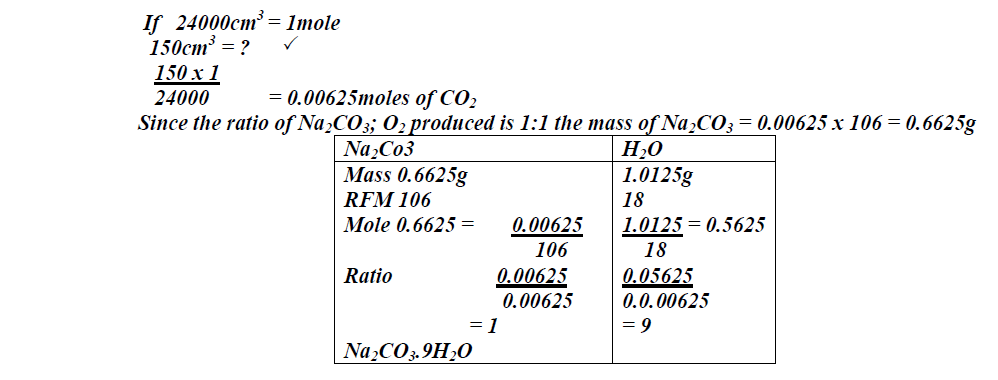
When 1.675g of hydrated sodium carbonate was reacted with excess hydrochloric acid, the volume carbon (IV) oxide gas obtained at room temperature and pressure was 150cm<sup>3</sup>). Calculate...
When exposed to air, crystals of hydrated sodium carbonate loses water of crystallizations;- i).Name - Tutorke

10-hydrate Sodium Carbonate Na2co3.10h2o Ar Grade - Buy Sodium Carbonate Na2co3.10h2o,10-hydrate Sodium Carbonate,Sodium Carbonate Decahydrate Ar Grade Product on Alibaba.com