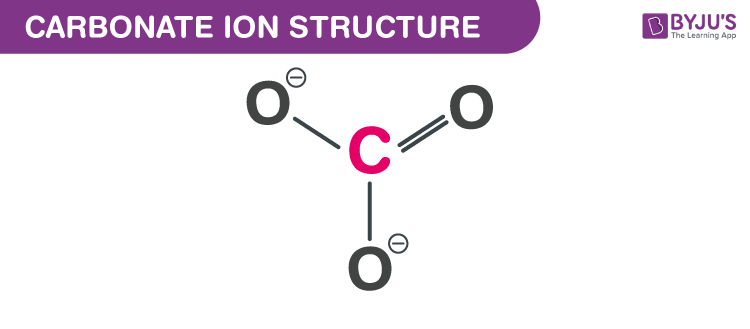
write the chemical formula of the sodium carbonate using Criss cross method please don't ignore the Criss - Brainly.in
![SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- ( SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- (](https://cdn.numerade.com/ask_previews/d76bf241-7907-4c05-a2e2-3a50f51ffda2_large.jpg)
SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- (

a) Define 'formula unit' of an ionic compound. What is the formula unit of (i) sodium chloride, and (ii) magnesium chloride ? (b) Calculate the formula masses of the following compounds : (
 \\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ... The molecular formula of sodium carbonate is:(A) \\[{\\text{NaC}}{{\\text{O}}_3}\\](B) \\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ...](https://www.vedantu.com/question-sets/2e37659b-297f-488b-bc60-a7c4facc2cff8373828392292757040.png)
The molecular formula of sodium carbonate is:(A) \\[{\\text{NaC}}{{\\text{O}}_3}\\](B) \\[\\left( {{\\text{N}}{{\\text{H}}_4}} \\right){\\text{S}}{{\\text{O}}_4}\\](C) \\[{\\text{N}}{{\\text{a}}_2}{\\text{C}}{{\\text{O}}_3}\\](D) \\[{\\left( {{\\text{N ...

Reactions of Carboxylic Acids (4.8.3) | Edexcel International A Level Chemistry Revision Notes 2017 | Save My Exams
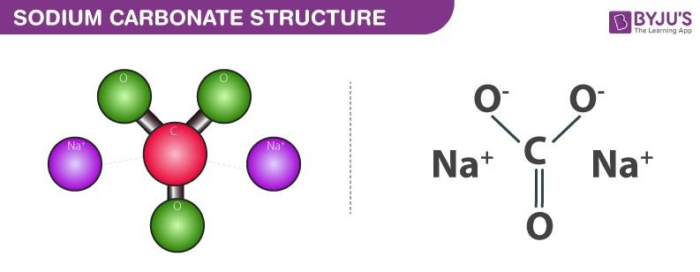








![The formula of sodium carbonate is \\[N{{a}_{2}}C{{O}_{3}}\\].A.TrueB.False The formula of sodium carbonate is \\[N{{a}_{2}}C{{O}_{3}}\\].A.TrueB.False](https://www.vedantu.com/question-sets/554ac5da-4846-4ac6-8fda-bb61c23ac5f425910065637539788.png)