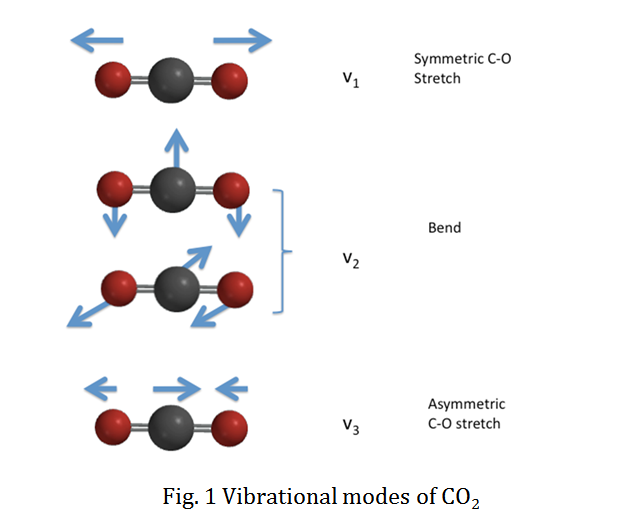
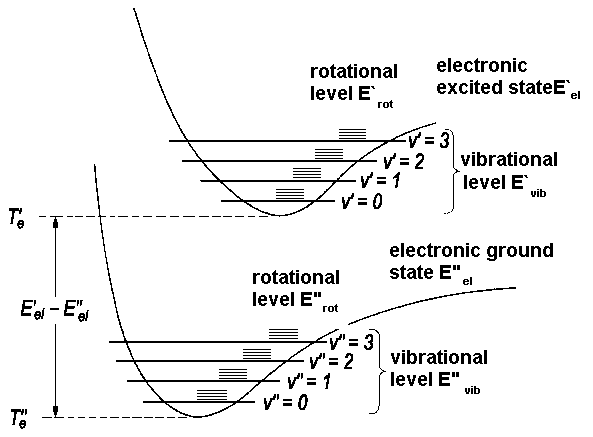
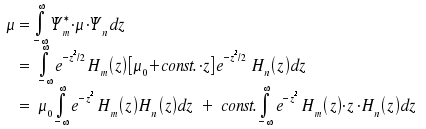SOLVED: Question 2 10 pts A diatomic molecule can be characterized by its vibrational quantum number (n) and rotational quantum number (J): The selection rules for an infrared vibrational transition of a

Symmetry selection rules for vibrational excitation by resonant electron impact and a unified treatment of vibronic coupling between resonances and to the continuum: A complete symmetry analysis of vibrational excitation in benzene
and similarly for Ψf. In the case of a vibrational transition, and the intensity becomes → No vibrational spectra! → Vibrat