What precipitate will form when aqueous solutions of sodium carbonate calcium Na_2CO_3 and calcium chloride CaCl_2 are mixed? | Socratic
Sodium Nitrate Crystal Sodium Carbonate, PNG, 610x898px, Sodium Nitrate, Body Jewelry, Calcium Nitrate, Chemical Compound, Chemistry
What is the molecular, complete ionic, and net ionic equation for calcium nitrate and sodium carbonate? - Quora

What happens when aqueous solutions of calcium chloride and of sodium carbonate are mixed? | Socratic
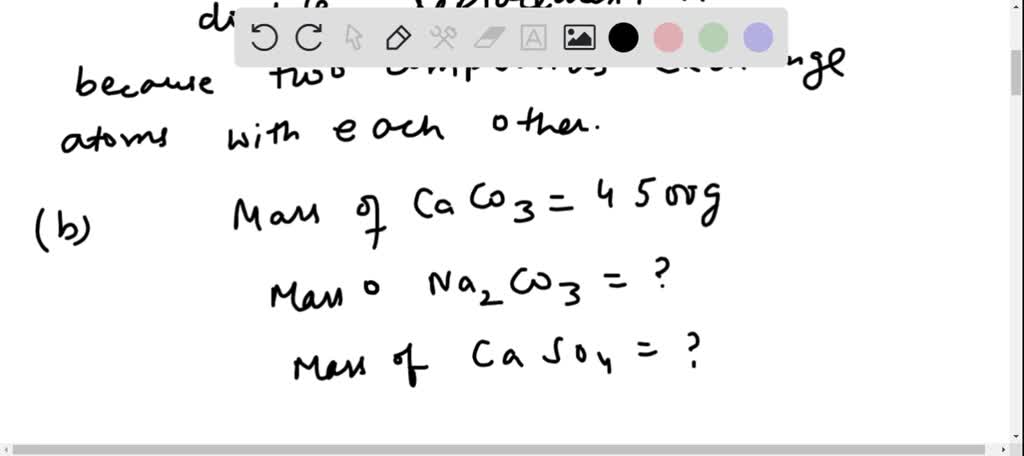
SOLVED: Calcium nitrate reacts with sodium carbonate to precipitate solid calcium carbonate: Ca(NO3)2(aq) + Na2CO3(aq) ? CaCO3(s) + NaNO3(aq) Balance the chemical equation. How many grams of Na2CO3 are needed to react
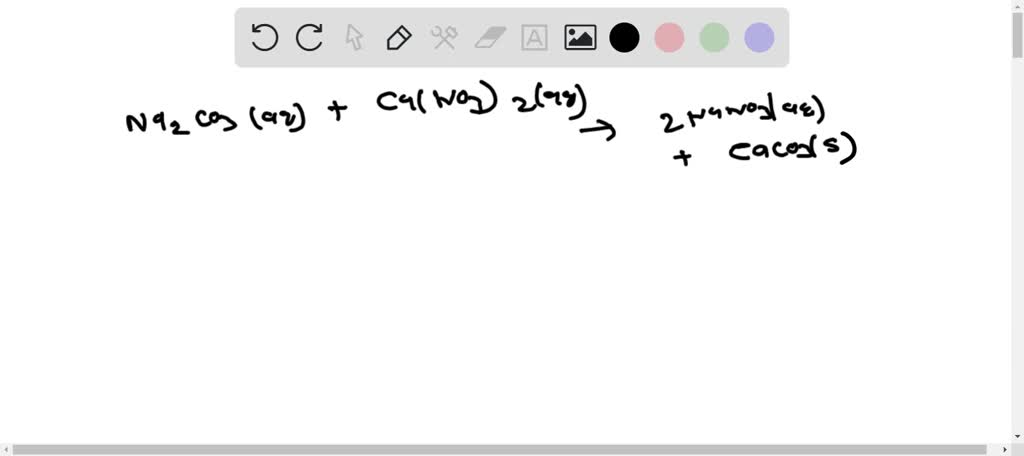
SOLVED: Write molecular, ionic and net ionic equations for following reactions Calcium Nitrate with Sodium Carbonate Calcium Nitrate with Sodium Hydroxide Calcium Nitrate with Trisodium Phosphate