Write a balanced chemical equation for the following chemical reaction : Magnesium burns in oxygen - YouTube

Empirical formula of magnesium oxide Background The empirical formula of a compound is the simplest value of the ratio of atoms of each element in the. - ppt download
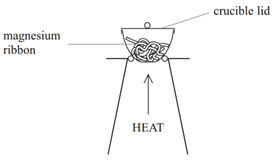
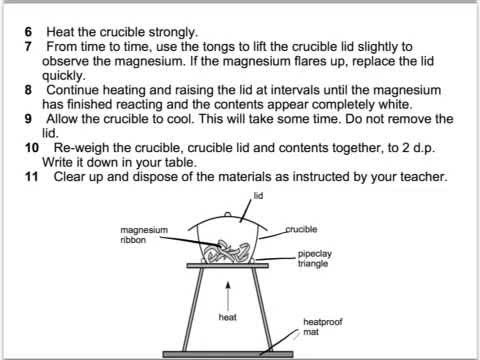
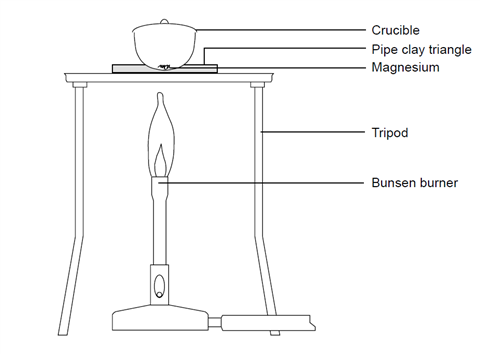
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry

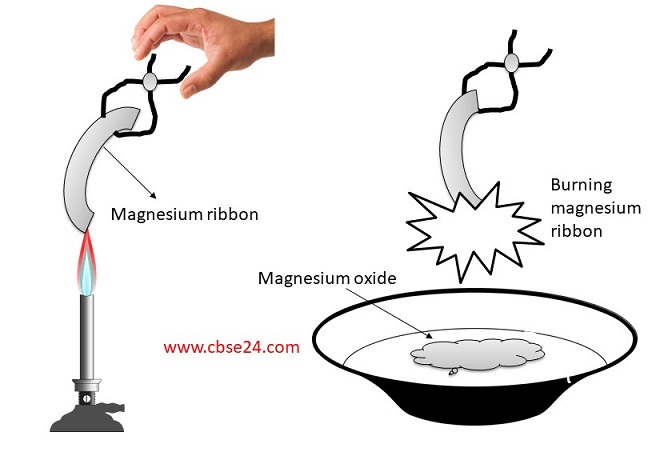
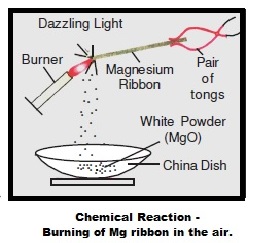
A magnesium ribbon is burnt in oxygen to give a white compound 'X' accompanied by emission of light. If the burning ribbon is now placed in an atmosphere of nitrogen, it continues

What is a redox reaction? When a magnesium ribbon burns in air with a dazzling flame and forms - YouTube

SOLVED: Actual formula of magnesium oxide (based on charge of Mg and " 0: ZMg 2 mc (s) +02 P 275 Theoretical mass % of Mg based n the actual formula of

a magnesium ribbon is burnt is oxygen to give a white compound x accompanied by emissionof light.write - Brainly.in

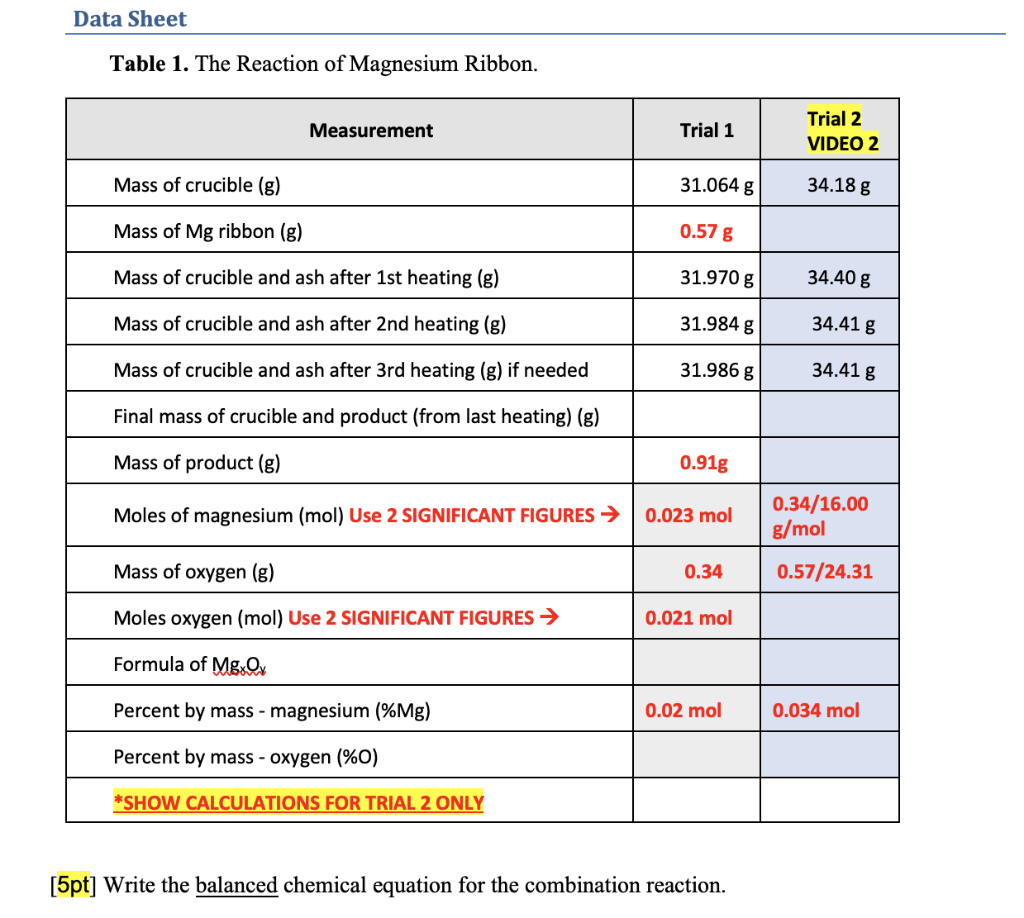
SOLVED: Table 1. The Reaction of Magnesium Ribbon Measurement Trial Mass cruciblelg 31.06 Mass of Mg ribbonlg Mass of crucible and Mg ribbonlg) 31.63 Mass crucible and procuct (after heating) (g 31.97

PDF) Experimentally Determining of the Empirical Formula of Magnesium Oxide | Folk Narongrit - Academia.edu